Snowshoe Hares Can't Outrun Climate Change

TL;DR: Bacteria build sophisticated biofilm communities with plumbing networks, specialized neighborhoods, and chemical communication systems. These microbial cities make infections up to 1,000 times harder to treat, but new strategies targeting their architecture could transform medicine.

Imagine a world where the most sophisticated urban planners aren't human. They don't have brains, blueprints, or even a single neuron between them. They're bacteria, and they've been building cities for billions of years before we ever stacked one brick on top of another. These microscopic metropolises come complete with plumbing systems, specialized neighborhoods, chemical communication networks, and defensive walls that make medieval fortifications look flimsy. And right now, they're probably thriving on your kitchen sponge, inside a hospital catheter, or deep within someone's lungs, where they're winning a war against our best antibiotics.
For most of modern medicine's history, scientists studied bacteria as lone wolves, isolated cells swimming freely in lab dishes. That mental model shaped everything, from how we designed antibiotics to how we understood infections. But starting in the late 20th century, researchers began to realize that this picture was fundamentally wrong. In nature, the vast majority of bacteria don't float around solo. They build structured communities called biofilms, anchored to surfaces and encased in a self-produced matrix of sugars, proteins, and DNA.
The numbers are staggering. According to estimates from the National Institutes of Health, 65 to 80 percent of all bacterial infections involve biofilms. That plaque on your teeth? A biofilm. The persistent lung infections that plague cystic fibrosis patients? Biofilm. The stubborn infections that form on hip replacements and heart valves? Also biofilms. And here's what makes them truly terrifying for doctors: bacteria living inside these communities can be up to 1,000 times more resistant to antibiotics than the same bacteria floating freely in your bloodstream.
Understanding the architecture of these microbial cities isn't just academically fascinating. It could be the key to solving one of medicine's most urgent problems.
The comparison between biofilms and human cities isn't just a cute metaphor. It's structurally accurate in ways that keep surprising researchers. Just as the printing press transformed how humans coordinated across distances, the discovery of quorum sensing, the chemical language bacteria use to coordinate behavior, transformed how microbiologists think about bacterial life.
Quorum sensing was first described in the bioluminescent marine bacterium Vibrio fischeri in the 1970s. Scientists noticed that these bacteria only produced light when enough of them gathered together. They were essentially taking a census, releasing signaling molecules called autoinducers and waiting until the concentration hit a threshold before switching on group behaviors. Since then, quorum sensing has been identified as a universal coordinator of biofilm development across hundreds of species.
Think of it as the internet of the bacterial world. When enough cells detect a quorum, they collectively switch on genes for building the extracellular matrix, the structural material that holds everything together. Pseudomonas aeruginosa, one of the most studied biofilm formers, uses three distinct quorum sensing circuits to orchestrate its community behavior, a level of regulatory complexity that rivals multicellular organisms.
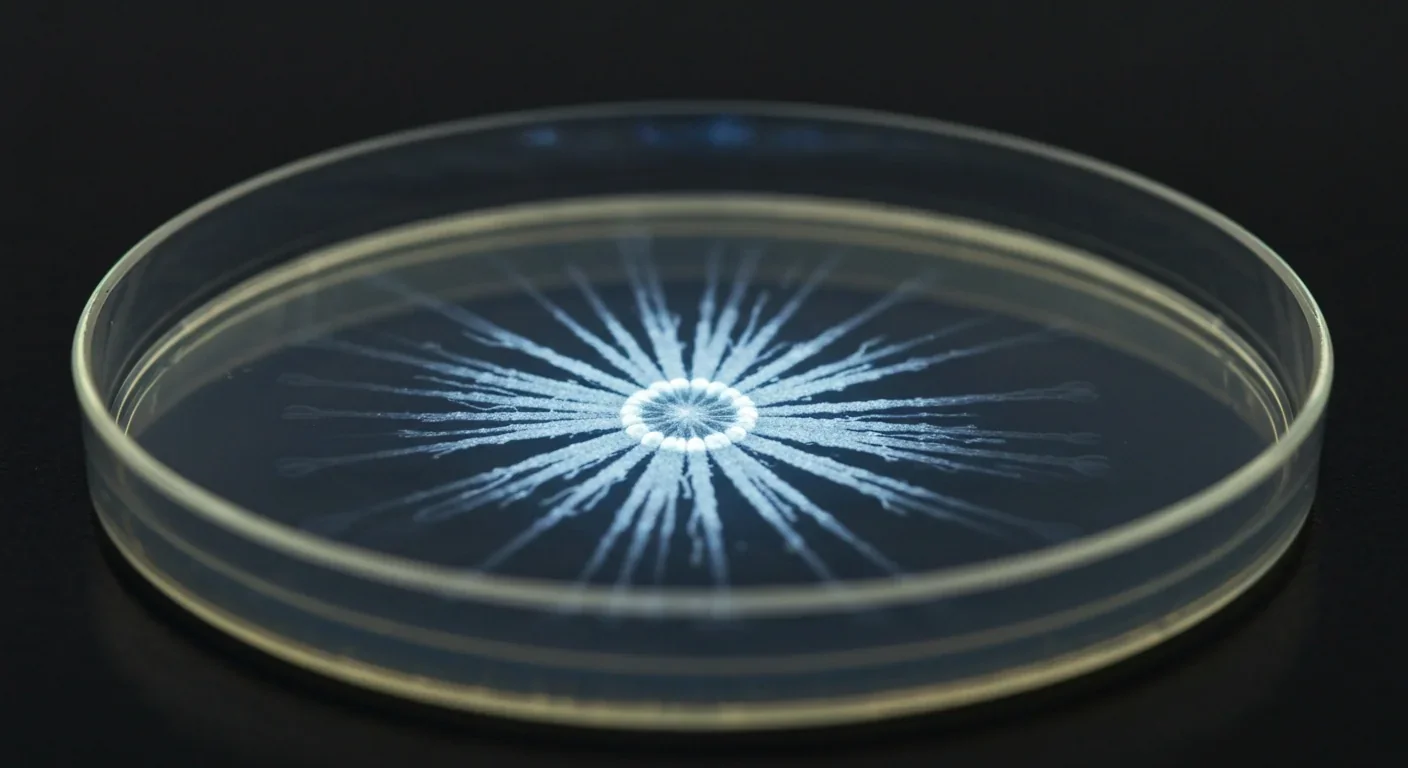
This coordination isn't new. Fossil evidence suggests bacteria have been forming biofilm-like communities for over 3 billion years. They were building cooperative societies long before the first multicellular organism appeared. In a real sense, bacteria invented urban planning.
So what does this city actually look like under a microscope? The answer has become dramatically clearer thanks to confocal laser scanning microscopy, which lets researchers build three-dimensional maps of living biofilms without destroying them.
The first thing you notice is the matrix. This isn't a random blob of goo. The extracellular matrix typically constitutes over 90 percent of a biofilm's dry mass, with the actual bacterial cells making up less than 10 percent. It's as if the buildings in a city took up 90 percent of the space and the people only 10. The matrix is composed of at least three major biopolymers: exopolysaccharides, extracellular proteins, and extracellular DNA. Each plays a distinct role. Polysaccharides provide the scaffold, a hydrogel-like framework that retains water and gives the biofilm its shape. Proteins like TasA in Bacillus subtilis act as structural reinforcement, stabilizing the matrix and enabling division of labor that benefits the whole community.
And eDNA serves as a sticky adhesive that binds cells together while also, remarkably, functioning as a medium for horizontal gene transfer, letting bacteria share genetic information including antibiotic resistance genes.
The biofilm matrix makes up over 90% of the structure's dry mass. The bacteria themselves are less than 10%. It's as if a city's buildings occupied 90% of the space and the residents only 10%.
Recent research has revealed something even more surprising about the DNA component. In polymicrobial periodontal biofilms, the extracellular DNA is predominantly host-derived, originating from the body's own immune cells. The immune system's attempts to fight the infection actually provide building materials that strengthen the biofilm's defenses. Your body's own weapons become the enemy's fortification walls.
Perhaps the most striking city-like feature of biofilms is their internal plumbing. Mature biofilms contain water channels that function analogously to primitive circulatory systems, delivering nutrients from the surface to cells buried deep in the interior and flushing out metabolic waste.
Research using fluorescent latex microbeads has shown that these channels form extensive fissure networks penetrating even through biofilms more than 4,000 micrometers thick. That's four millimeters of living architecture, threaded with conduits so fine that only molecules smaller than about 2.2 nanometers can reach the deepest layers. Bacterial swimmers actually create pores in the matrix, forming these rudimentary circulation systems through their own movement.
But these channels aren't simple passive pipes. Studies of E. coli biofilms have found that oxygen concentration changes dramatically along the length of transport channels, contradicting earlier assumptions that they were completely open conduits. Instead, localized growth layers at channel boundaries create controlled microenvironments, more like a sophisticated irrigation system with built-in flow regulation than a basic drainage ditch.
The functional result is remarkable. These channel networks maintain pH gradients ranging from 4 to 7 across different zones, creating distinct chemical neighborhoods within a single biofilm. Different bacteria thrive in different pH conditions, so these gradients effectively zone the city into specialized districts.
Here's where the city metaphor gets truly uncanny. Genetically identical bacteria within a biofilm don't all behave the same way. They differentiate into specialized roles driven by gradients of oxygen, nutrients, and signaling molecules. Some become matrix producers, pumping out the polysaccharides and proteins that build and maintain the infrastructure. Others become nutrient transport workers. Still others become persisters, dormant sleeper cells that serve as the ultimate insurance policy.
This division of labor has real costs and benefits. Research on B. subtilis biofilms has shown that matrix components like EPS and TasA are costly public goods that facilitate division of labor optimized at the population level. Some cells pay the metabolic cost of building infrastructure while others free-ride. But the community has evolved anti-cheating mechanisms.
"The exopolysaccharide (EPS) and protein components (TasA) of B. subtilis biofilms are costly public goods that facilitate the division of labour that is optimal at the genotypic level."
- B. subtilis biofilm research, Researcher.life
When non-producers try to exploit the system, the biofilm can deploy bacteriophage-mediated responses that keep freeloaders in check, essentially a bacterial version of policing.
Even across different species, this cooperation holds. Pseudomonas syringae, which infects plants, divides its population into subgroups with distinct roles during infection. Multi-species biofilms show emergent properties, including enhanced antibiotic resistance and metabolic capabilities that no single species possesses alone.
This brings us to the medical crisis. Biofilms aren't just scientifically interesting. They're a massive clinical problem. Biofilm-related infections cost healthcare systems billions annually and are a leading cause of hospital-acquired infections. They're implicated in up to 60 percent of chronic wounds and are central to the pathogenesis of diabetic foot ulcers and burn infections.

The problem is architectural. Conventional antibiotics were designed to kill free-floating bacteria by targeting growth-dependent processes: cell wall synthesis, DNA replication, protein production. But dormant persister cells have low metabolic activity and simply don't use these processes at the rate antibiotics expect.
These persisters comprise roughly 1 percent of the biofilm population, but they're the ones that matter most. After an antibiotic course wipes out the active cells, persisters wake up and rebuild the entire community.
Persister cells make up just 1% of a biofilm's population, but they're the reason infections keep coming back. These dormant sleeper cells survive antibiotic treatment and rebuild the entire community from scratch.
The matrix itself compounds the problem. Extracellular DNA within the matrix binds positively charged antibiotics like aminoglycosides, physically sequestering them before they can reach bacterial cells. Meanwhile, enzymes embedded in the matrix can inactivate beta-lactam antibiotics before they penetrate deep enough to be effective.
For cystic fibrosis patients, this is a matter of life and death. Pseudomonas aeruginosa biofilms in CF lungs are a primary cause of morbidity and mortality, establishing chronic infections that can persist for decades despite aggressive antibiotic therapy.
The good news is that understanding biofilm architecture has opened entirely new therapeutic avenues. Instead of trying to kill bacteria directly, researchers are now targeting the city's infrastructure.
Matrix-degrading enzymes represent one of the most promising approaches. Dispersin B, which breaks down PNAG polysaccharides, can eradicate up to 95 percent of mature S. aureus biofilms when combined with vancomycin. DNase enzymes that chew through the eDNA scaffold are showing similar promise in combination therapies.
Quorum sensing inhibitors attack the communication system. If you can jam the chemical signals bacteria use to coordinate, you can prevent the biofilm from ever forming properly. Thymoquinone has demonstrated significant reduction in QS gene expression and biofilm integrity in P. aeruginosa. Other researchers are exploring quorum quenching enzymes like lactonases that degrade the signaling molecules themselves.
"Bacterial persisters represent a promising target for biotherapeutic intervention against chronic biofilm infections."
- Kim W. et al., IntechOpen
For persister cells, scientists are developing strategies to either wake them up or kill them while dormant. Metabolic reactivation approaches use fatty-acid signaling molecules to shift persisters into an active metabolic state where conventional antibiotics can reach them. Synthetic retinoids like CD437 and CD1530 take the opposite approach, disrupting persister cell membranes directly without requiring metabolic activity.
Nitric oxide has emerged as a versatile weapon. NO-releasing hydrogel wound dressings have achieved over 99 percent reduction in MRSA biofilm in wound models. And engineered nanoparticles with high surface-to-volume ratios can penetrate both the surface EPS and microchannels, acting as dual-anchor delivery systems.
The story isn't only about destruction. Biofilm architecture is inspiring engineers and materials scientists in unexpected ways.
Biofilms are already being harnessed in wastewater treatment, where their natural ability to break down organic pollutants makes them invaluable. Industrial applications range from bioremediation to microbial fuel cells, and researchers are studying how biofilm channel networks could inspire self-fluidic microdevices for drug delivery.
Computational models are now simulating biofilm matrix architecture at multiple scales, helping researchers understand how the interplay between individual cell behavior and collective structure produces such robust, adaptive communities. These models are revealing principles of self-organization that could apply to everything from designing resilient materials to understanding how cities themselves might better manage resource distribution.
Within the next decade, you'll likely encounter biofilm-targeted therapies in clinical practice. The research pipeline is moving from laboratory curiosity to clinical reality, with combination approaches attacking multiple aspects of biofilm architecture simultaneously.
The deeper lesson here goes beyond medicine. Bacteria figured out collective problem-solving, resource distribution, infrastructure engineering, and defensive architecture billions of years before we did. They solved the challenge of cooperative living at scales we're only beginning to appreciate. The question isn't whether we can learn from their designs. It's whether we can learn fast enough to stay ahead of them.

Solar sail spacecraft navigate the solar system by tacking on sunlight, angling reflective sheets to redirect photon pressure just as sailboats tack against the wind. Missions like IKAROS and LightSail 2 have proven the physics works, and next-generation designs could enable interstellar travel.

Metformin, a cheap diabetes drug taken by 150 million people, may slow aging by mildly stressing cells through mitochondrial complex I inhibition, triggering protective AMPK pathways. The landmark TAME trial is now testing this in humans, potentially making aging itself an FDA-treatable condition.

Scientists have identified 16 climate tipping elements that can trigger each other in cascading chain reactions. New research shows five may already be at risk at current warming, and crossing just one threshold can double the number of systems that collapse.

The cheerleader effect is a proven cognitive bias where people look more attractive in groups because the brain automatically averages faces, smoothing out individual flaws. Research shows the sweet spot is 3-5 people, it works for all genders, and it has real implications for dating apps and social media strategy.

Snowshoe hares change coat color based on day length, not snow. As climate change shortens snow seasons, white hares stand out against brown landscapes, increasing predation by 7% per week. Some populations carry a borrowed gene for staying brown, offering a potential genetic lifeline.

Care workers earn poverty-level wages despite performing essential labor worth trillions globally. Historical gendering of domestic work, flawed economic models, and systemic racism entrench this undervaluation, but evidence from Nordic countries and union organizing shows that treating care as infrastructure produces massive economic returns.

Google, IBM, and every major quantum computing company converged on the transmon qubit because it offered the best trade-off between noise immunity and manufacturability. Flux qubits survive in D-Wave's annealers, and hybrid fluxonium designs may soon challenge transmon dominance.